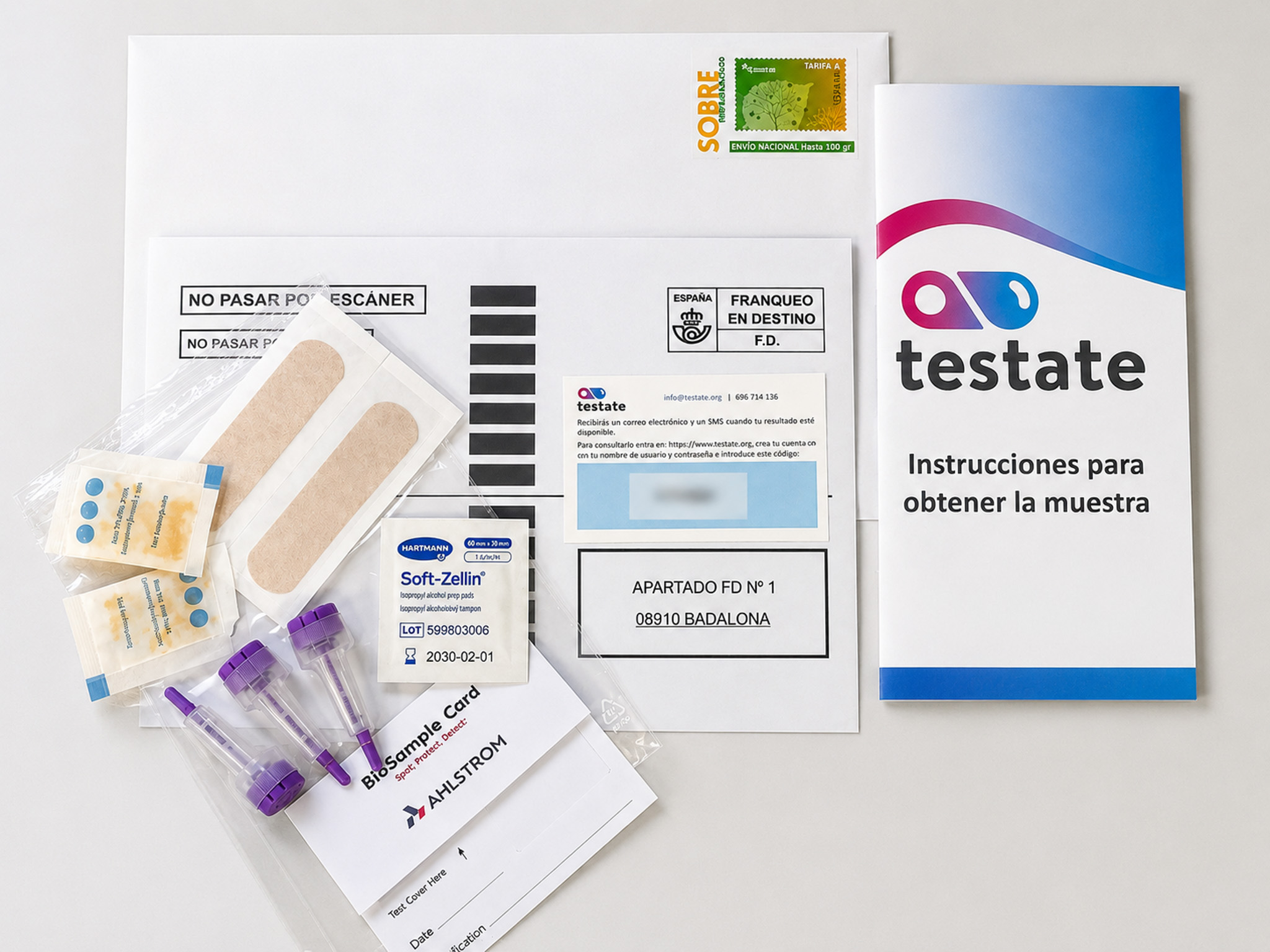
CEEISCAT advances Pan-Téstate to strengthen online screening for HIV and viral hepatitis among GBMSM and trans women in Spain
CEEISCAT, a research group at IGTP, is developing Pan-Téstate, an online self-sampling program aimed at improving early screening for HIV and viral hepatitis among GBMSM and trans women in Spain. Using dried blood spot samples collected at home, the initiative seeks to reduce barriers to testing and improve access to early diagnosis and prevention.